Today, there are two technologies available on the portable XRF spectrometer market: analyzers with an X-ray tube and analyzers with a radioactive source.
To sort metals at a scrap yard, an XRF device with an X-ray tube such as the FEnX-T is used, while to detect lead in paint, an XRF device with a radioactive source such as the FEnX2 is used.
In this article, we explain the difference between the two technologies and why a spectrometer with a radioactive source is used to perform lead diagnostics.
First, let’s review the concepts of atoms, electron shells, and XRF technology.
The atom and electron shells
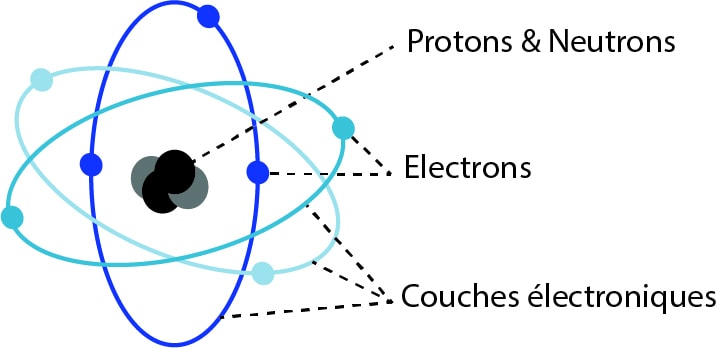
An atom consists of a central nucleus and electrons that orbit the nucleus in different electron shells or energy orbits.
Each shell has a corresponding energy level.
The atom can also be illustrated as follows, with a nucleus and the K, L, and M layers:
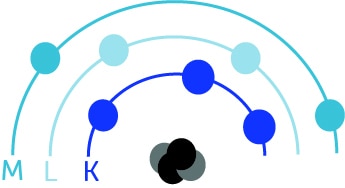
The layer closest to the nucleus is the K layer.
Electrons constantly seek to remain in their initial electron layer (K, L, or M).
To remain in orbit in its layer, an electron needs binding energy. And the closer the layer is to the nucleus, the more energy is required to remain there.
The heavier an atom is, the more electrons it has and the more filled layers it has.
Lead, a heavy atom
The lead atom is considered a heavy atom because it has many electrons and many filled shells. It ranks 82nd in the periodic table.
Focus on the K and L shells of lead
In the case of the lead atom, an electron in a K shell will need between 74 and 85 keV of energy to remain in orbit.
An electron in an L shell will need between 10 and 12 keV of energy to remain in orbit.
Now that the concepts of atoms and shells have been clarified, we can return to XRF technology.
Detecting an element in a sample with XRF technology
To detect an element in a sample—lead in paint, for example—we seek to remove electrons from their shells using radiation emitted by the spectrometer.
Why remove these electrons?
When an electron is removed from its shell, a hole is created. The atom is then said to be unstable.
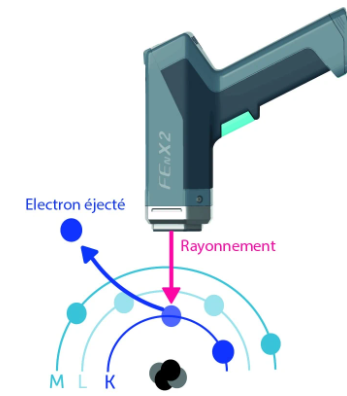
The inner layers will attempt to reorganize themselves by recovering electrons from the upper layers to fill the gap created and thus return to a stable state. This movement of electrons between the layers causes energy to be emitted in the form of X-rays, known as X-ray fluorescence. This is the difference in energy between the two layers.
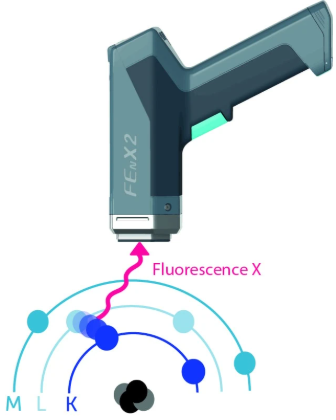
These energies will be captured by the spectrometer detector: they will create an electrical signal that is shaped to generate a spectrum.
The K, L, and M spectral lines correspond to the collection of different X-ray fluorescence emissions from the material: an electron that is stripped from the K layer will contribute to forming the K spectral line.
To strip an electron from its layer, energy greater than that of the layer is required. In Figure 1, the energy emitted by the device must be greater than that of the K layer.
The concepts of atoms, spectral lines, and X-ray fluorescence are reviewed. The difference between an X-ray tube device and a source device can be explained.
What is the difference between an X-ray tube device and a radioactive source device?
Currently, an XRF spectrometer can contain an X-ray tube with an energy of up to 50 kV. This is the case, for example, with the FEnX-T.
An XRF spectrometer designed to analyze lead in paint may contain a radioactive cadmium-109 source (FEnX) or a cobalt-57 source (Pb200i). The maximum energy emitted by a cadmium-109 source is approximately 88 keV, while that of a cobalt source is 120 keV.
The difference is therefore the energy: the emission energy of a source spectrometer is greater than that of an X-ray tube spectrometer.
Thus, returning to the example of lead, in order to remove electrons from the K electron shell, it is essential that the energy emitted by the spectrometer be greater than the energy of the K lines of lead (between 74 keV and 85 keV).
But why analyze the K lines to detect lead in paint?
The decree of August 19, 2011, on the assessment of lead exposure risk stipulates that “Lead concentration measurements in coatings shall be carried out for each diagnostic unit. They shall be performed using a portable X-ray fluorescence device capable of analyzing at least the K line of the fluorescence spectrum emitted in response to lead. “
In a 2005 opinion from the AFSSE (French Agency for Environmental Health Safety) concerning the detection of lead in old paint, it is explained that ”The measurement of the X-ray fluorescence spectrum of an element may be affected […] This is the case when the lead is located deep down or covered by one or more layers containing one or more interfering elements.”
In concrete terms: If a layer of paint containing lead is covered by wallpaper (or any other coating of significant thickness), it may be important to analyze the K lines.
If only the L layers are analyzed, the corresponding X-ray fluorescence could be altered or obscured by the coating covering the lead paint layer.
The spectral analysis resulting solely from the L lines could therefore be distorted.
With a tube XRF spectrometer, there is a risk of giving a false negative, i.e., the result would show that there is no lead when in fact there is lead under the wallpaper (or under the coating of significant thickness).
This is why French regulations require the use of a spectrometer capable of analyzing the K line of the fluorescence spectrum emitted in response to lead.

